where z is known as the stoichiometric coefficient for the oxidizer (air)
Chapter 11: Combustion
(Thanks
to David
Bayless for his assistance in writing
this section)
Introduction - Up to this point the heat Q in all problems and examples was either a given value or was obtained from the First Law relation. However in various heat engines, gas turbines, and steam power plants the heat is obtained from combustion processes, using either solid fuel (e.g. coal or wood). liquid fuel (e.g. gasolene, kerosine, or diesel fuel), or gaseous fuel (e.g. natural gas or propane).
In this chapter we introduce the chemistry and thermodynamics of combustion of generic hydrocarbon fuels - (CxHy), in which the oxydizer is the oxygen contained in atmospheric air. Note that we will not cover the combustion of solid fuels or the complex blends and mixtures of the hydrocarbons which make up gasolene, kerosene, or diesel fuels.
Atmospheric Air contains approximately 21% oxygen (O2) by volume. The other 79% of "other gases" is mostly nitrogen (N2), so we will assume air to be composed of 21% oxygen and 79% nitrogen by volume. Thus each mole of oxygen needed to oxidize the hydrocarbon is accompanied by 79/21 = 3.76 moles of nitrogen. Using this combination the molecular mass of air becomes 29 [kg/kmol]. Note that it is assumed that the nitrogen will not normally undergo any chemical reaction.
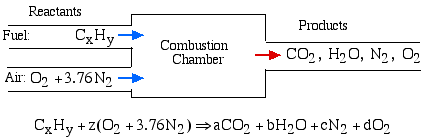
The Combustion Process - The basic combustion process can be described by the fuel (the hydrocarbon) plus oxydizer (air or oxygen) called the Reactants, which undergo a chemical process while releasing heat to form the Products of combustion such that mass is conserved. In the simplest combustion process, known as Stoichiometric Combustion, all the carbon in the fuel forms carbon dioxide (CO2) and all the hydrogen forms water (H2O) in the products, thus we can write the chemical reaction as follows:
where z is known as the stoichiometric coefficient for the oxidizer (air)
Note that this reaction yields five unknowns: z, a, b, c, d, thus we need five equations to solve. Stoichiometric combustion assumes that no excess oxygen exists in the products, thus d = 0. We obtain the other four equations from balancing the number of atoms of each element in the reactants (carbon, hydrogen, oxygen and nitrogen) with the number of atoms of those elements in the products. This means that no atoms are destroyed or lost in a combustion reaction.
|
Element |
Amount in reactants |
= |
Amount in Products |
Reduced equation |
|
Carbon (C) |
x |
|
a |
a = x |
|
Hydrogen (H) |
y |
|
2b |
b = y/2 |
|
Oxygen (O) |
2z |
|
2a+b |
z = a + b/2 |
|
Nitrogen (N) |
2(3.76)z |
|
2c |
c = 3.76z |
Note that the water formed could be in the vapor or liquid phase, depending on the temperature and pressure of the combustion products.
As an example consider the stoichiometric combustion of methane (CH4) in atmospheric air. Equating the molar coefficients of the reactants and the products we obtain:
Theoretical Air and Air-Fuel Ratio -The minimum amount of air which will allow the complete combustion of the fuel is called the Theoretical Air (also referred to as Stoichiometric Air). In this case the products do not contain any oxygen. If we supply less than theoretical air then the products could include carbon monoxide (CO), thus it is normal practice to supply more than theoretical air to prevent this occurrence. This Excess Air will result in oxygen appearing in the products.
The standard measure of the amount of air used in a combustion process is the Air-Fuel Ratio (AF), defined as follows:
Thus considering only the reactants of the methane combustion with theoretical air presented above, we obtain:
Solved Problem 11.1 - In this problem we wish to develop the combustion equation and determine the air-fuel ratio for the complete combustion of n-Butane (C4H10) with a) theoretical air, and b) 50% excess air.
Analysis of the Products of Combustion - Combustion always occurs at elevated temperatures and we assume that all the products of combustion (including the water vapor) behave as ideal gases. Since they have different gas constants, it is convenient to use the ideal gas equation of state in terms of the universal gas constant as follows:
In the analysis of the products of combustion there are a number of items of interest:
1) What is the percentage volume of specific products, in particular carbon dioxide (CO2) and carbon monoxide (CO)?
2) What is the dew point of the water vapor in the combustion products? This requires evaluation of the partial pressure of the water vapor component of the products.
3) There are experimental methods of volumetric analysis of the products of combustion, normally done on a Dry Basis, yielding the volume percentage of all the components except the water vapor. This allows a simple method of determining the actual air-fuel ratio and excess air used in a combustion process.
For ideal gases we find that the mole fraction yi
of the i'th component in a mixture of gases at a specific pressure P
and temperature T is equal to the volume fraction of that component.
Since from the molar ideal gas relation: P.V = N.Ru.T,
we have:
Furthermore, since the sum of the component volumes Vi must equal the total volume V, we have:
Using a similar approach we determine the partial pressure of a component using Dalton's Law of Partial Pressures:
Solved Problem 11.2 - In this problem Propane (C3H8) is burned with 61% excess air, which enters a combustion chamber at 25°C. Assuming complete combustion and a total pressure of 1 atm (101.32 kPa), determine a) the air-fuel ratio [kg-air/kg-fuel], b) the percentage of carbon dioxide by volume in the products, and c) the dew point temperature of the products.
Solved Problem 11.3 - In this problem Ethane (C2H6) is burned with atmospheric air, and the volumetric analysis of the dry products of combustion yields the following: 10% CO2, 1% CO, 3% O2, and 86% N2. Develop the combustion equation, and determine a) the percentage of excess air, b) the air-fuel ratio, and c) the dew point of the combustion products.
The First Law Analysis of Combustion - The main purpose of combustion is to produce heat through a change of enthalpy from the reactants to the products. From the First Law equation in a control volume, ignoring kinetic and potential energy changes and assuming no work is done, we have:
where the summations are taken over all the products (p) and the reactants (r). N refers to the number of moles of each component and h [kJ/kmol] refers to the molar enthalpy of each component.
Since there are a number of different substances involved we need to establish a common reference state to evaluate the enthalpy, the common choice being 25°C and 1 atm which is normally denoted with a superscript o. Prof. S. Bhattacharjee of the San Diego State University has developed a web based expert system at <www.thermofluids.net> called TEST (The Expert System for Thermodynamics) in which he has included a set of ideal gas property tables all based on the enthalpy ho = 0 at this common reference. We have adapted some of these tables specifically for this section, and these can be found in the following link:
Combustion Molar Enthalpy Tables
As an example, consider again the complete combustion of Methane (CH4) with theoretical air:
Notice that in the reactants and the products of the above example we have basic elements O2 and N2 as well as compounds CH4, CO2, and H2O. When the compound is formed then the enthalpy change is called the Enthalpy of Formation, denoted hfo, and for our example:
|
Substance |
Formula |
hfo [kJ/kmol] |
|
Carbon dioxide |
CO2(g) |
-393,520 |
|
Water Vapor |
H2O(g) |
-241,820 |
|
Water |
H2O(l) |
-285,820 |
|
Methane |
CH4(g) |
-74,850 |
where (g) refers to gas and (l) refers to liquid.
The negative sign means that the process is Exothermic, i.e. heat is given off when the compound is formed. Note that the enthalpy of formation of basic elements O2 and N2 is zero.
Consider first the case in which there is sufficient heat transfer such that both the reactants and the products are at 25°C and 1 atm pressure, and that the water product is liquid. Since there is no sensible enthalpy change the energy equation becomes:
This heat (Qcv) is called the Enthalpy of Combustion or the Heating Value of the fuel. If the products contain liquid water then it is the Higher Heating Value (as in our example), however if the product contains water vapor then it is the Lower Heating Value of the fuel. The enthalpy of combustion is the largest amount of heat that can be released by a given fuel.
Adiabatic Flame Temperature - The opposite extreme of the above example in which we evaluated the enthalpy of combustion is the case of an adiabatic process in which no heat is released. This results in a significant temperature increase in the products of combustion (denoted the Adiabatic Flame Temperature) which can only be reduced by an increase in the air-fuel ratio.
Solved Problem 11.4 - Determine the adiabatic flame temperature for the complete combustion of Methane ( CH4) with 250% theoretical air in an adiabatic control volume.
This equation can only be solved by an iterative trial and error procedure using the tables of Sensible Enthalpy vs Temperature for all four components of the products - CO2, H2O, O2, and N2. A quick approximation to the adiabatic flame temperature can be obtained by assuming that the products consist entirely of air. This approach was introduced to us by Potter and Somerton in their Schaum's Outline of Thermodynamics for Engineers, in which they assumed all the products to be N2. We find it more convenient to use air assuming a representative value of the Specific Heat Capacity of Air: Cp,1000K = 1.142 [kJ/kg.K].
Thus summing all the moles of the products we have:
Using the tables of Sensible Enthalpy vs Temperature we evaluated the enthalpy of all four products at a temperature of 1280K. This resulted in a total enthalpy of 802,410 [kJ/kmol fuel], which is extremely close to the required value, thus justifying this approach.
Problem 11.5 - - Determine the adiabatic flame temperature for the complete combustion of Propane ( C3H8) with 250% theoretical air in an adiabatic control volume [T = 1300K].
______________________________________________________________________________________
![]()
Engineering Thermodynamics by Israel
Urieli is licensed under a Creative
Commons Attribution-Noncommercial-Share Alike 3.0 United States
License